Thus, the columns of the periodic table represent the potential shared state of these elements' outer electron shells that is responsible for their similar chemical characteristics. 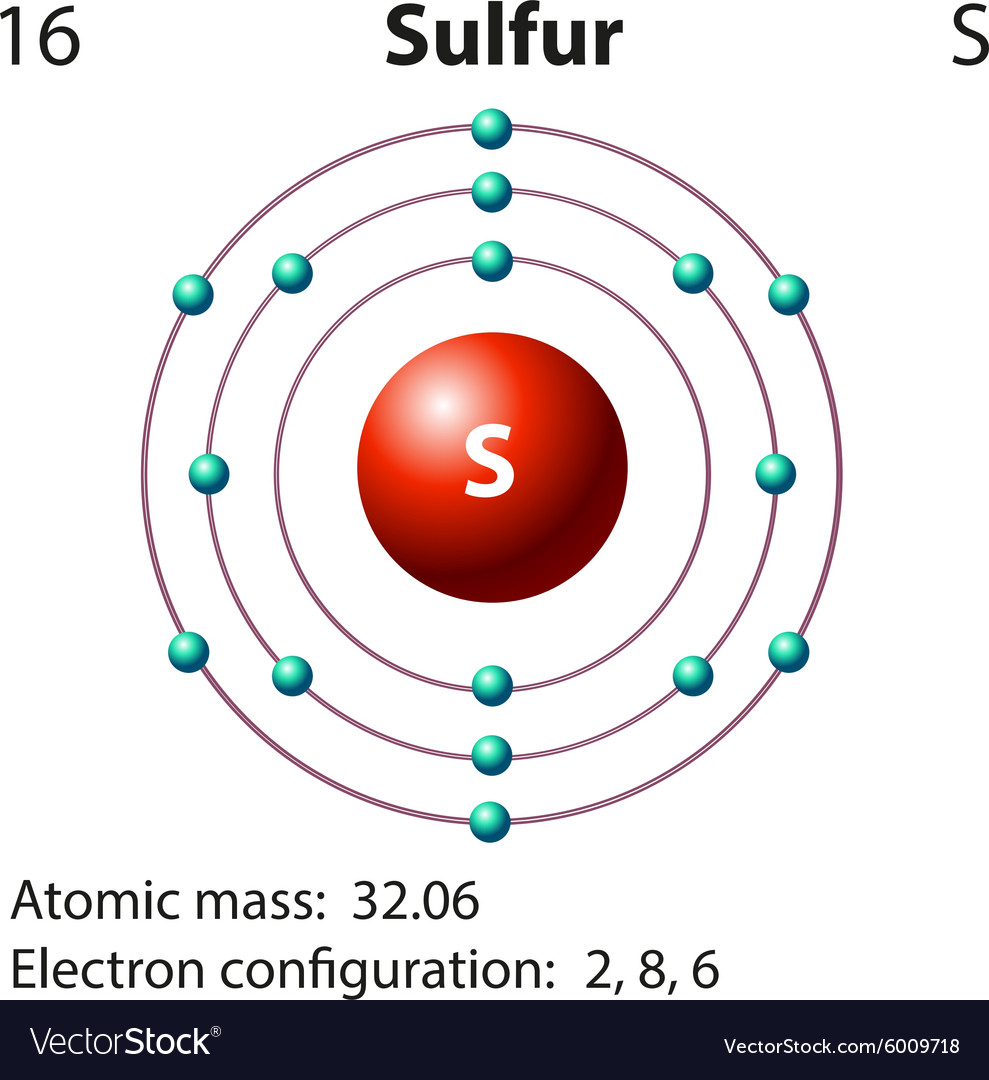
When an atom gains an electron to become a negatively-charged ion this is indicated by a minus sign after the element symbol for example, \(F^-\). Group 17 elements, including fluorine and chlorine, have seven electrons in their outermost shells they tend to fill this shell by gaining an electron from other atoms, making them negatively-charged ions. When an atom loses an electron to become a positively-charged ion, this is indicated by a plus sign after the element symbol for example, Na +. As a result of losing a negatively-charged electron, they become positively-charged ions. This means that they can achieve a stable configuration and a filled outer shell by donating or losing an electron. 
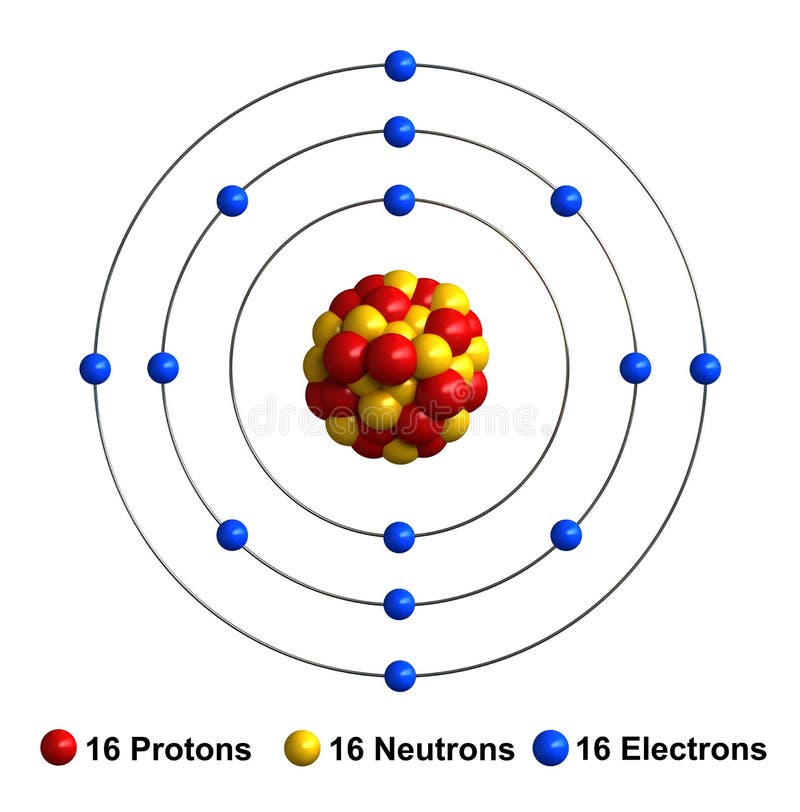
In comparison, the group 1 elements, including hydrogen (H), lithium (Li), and sodium (Na), all have one electron in their outermost shells. Their non-reactivity has resulted in their being named the inert gases (or noble gases). As shown in, the group 18 atoms helium (He), neon (Ne), and argon (Ar) all have filled outer electron shells, making it unnecessary for them to gain or lose electrons to attain stability they are highly stable as single atoms. With his model, Bohr explained how electrons could jump from one orbit to another only by emitting or absorbing energy in. As Bohr had noticed, the radius of the n 1 orbit is approximately the same size as an atom. The periodic table is arranged in columns and rows based on the number of electrons and where these electrons are located, providing a tool to understand how electrons are distributed in the outer shell of an atom. In Bohr’s model, radius an of the orbit n is given by the formula an h2n2 0 / 2, where 0 is the electric constant. Elements in other groups have partially-filled valence shells and gain or lose electrons to achieve a stable electron configuration.Īn atom may gain or lose electrons to achieve a full valence shell, the most stable electron configuration. A full valence shell is the most stable electron configuration. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. Understanding Bohr's model requires some knowledge of electromagnetic radiation (or light).īohr's key idea in his model of the atom is that electrons occupy definite orbitals that require the electron to have a specific amount of energy.\):īohr diagrams indicate how many electrons fill each principal shell. In 1913, the Danish physicist Niels Bohr proposed a model of the electron cloud of an atom in which electrons orbit the nucleus and were able to produce atomic spectra. These difficulties cast a shadow on the planetary model and indicated that, eventually, it would have to be replaced. Chlorine is neutral and its atomic number is 17, hence, the number of protons and electrons available for its Bohr diagram is also 17. Furthermore, Rutherford's model was unable to describe how electrons give off light forming each element's unique atomic spectrum. The Bohr model of Chlorine (Cl) is drawn with three electron shells, the first shell contains 2 electrons, the second shell contains 8 electrons and the third shell contains 7 electrons. If the electron circling the nucleus in an atom loses energy, it would necessarily have to move closer to the nucleus as it loses energy, and would eventually crash into the nucleus. This is, after all, how we produce TV signals. It was already known that when a charged particle (such as an electron) moves in a curved path, it gives off some form of light and loses energy in doing so. Unfortunately, there was a serious flaw in the planetary model. \): Niels Bohr with Albert Einstein at Paul Ehrenfest's home in Leiden (December 1925). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
